Sex-Biased Familial Aggregation of Darwin’s Tubercle in an Eastern Indian Tribal Population
P. Das1, A. R. Bandyopadhyay1
1Department of Anthropology, University of Calcutta, 35 Ballygunge Circular Road, Kolkata 700019, West Bengal, India.
*Corresponding author: abanthro@caluniv.ac.in
Abstract
Darwin’s tubercle (DT) is a minor auricular trait showing wide inter-population variation and non-Mendelian patterns of familial aggregation. This study examines sex-biased parent–offspring aggregation of DT in an Eastern Indian tribal population (Lodha PVTG), using 416 parent–offspring pairs. Significant associations were observed across all parental pathways (Chi² = 28.45–86.49; p < 0.001), with strongest aggregation in father–son pairs. The findings support autosomal expression with sex-specific penetrance and developmental modulation.
Keywords: Darwin’s tubercle; familial aggregation; ear morphology; developmental biology
Introduction
Darwin’s tubercle (DT) is a minor congenital thickening or projection located on the posterosuperior margin of the auricular helix and is widely regarded as a vestigial morphological trait in humans. Charles Darwin first described this feature as an atavistic remnant reflecting ancestral primate ear morphology, situating DT within the broader framework of evolutionary persistence and morphological variation (Darwin, 1871). Subsequent anatomical and anthropological investigations have reinforced this interpretation, recognizing DT as part of the natural spectrum of external ear variation across human populations (Hrdlička, 1933; Wood-Jones, 1931) Embryologically, the external ear develops from six auricular hillocks derived from the first and second pharyngeal arches, with critical morphogenesis occurring between the fifth and eighth weeks of gestation. Incomplete invagination or differential growth of the posterosuperior helix during this developmental window is considered the primary mechanism underlying DT expression (Porter, 2000; Sadler, 2019; Moore et al., 2020). This sensitivity of auricular development to minor perturbations provides a developmental basis for the considerable phenotypic variability observed in DT expression. Epidemiological studies report marked population-level variation in DT prevalence, ranging from approximately 10% in European populations to nearly 40% in Indian cohorts and exceeding 50% in certain Scandinavian samples (Porter, 2000; Bury et al., 2016). Indian anthropometric studies further document extensive diversity in auricular morphology, including helix configuration, lobule attachment, and tragus form, with DT occurring as part of this broader morphological continuum (Singh & Purkait, 2009; Deopa et al., 2013; Murgod et al., 2013). Although DT frequently presents bilaterally, asymmetrical expression is common, and most studies report no consistent association with sex or age. Despite its long-standing recognition, the genetic basis of DT remains incompletely understood. Early assumptions of simple autosomal dominant inheritance with incomplete penetrance have been challenged by family and twin studies demonstrating inconsistent transmission patterns, high expression among offspring of DT-negative parents, and discordance among monozygotic twins (Porter, 2000; McDonald, 2011). These observations suggest a more complex inheritance model involving multiple genetic and non-genetic influences. Accordingly, the present study situates DT within a polygenic and developmentally modulated framework, emphasizing its relevance to anthropological and forensic research.
This study aims to delineate inheritance patterns of Darwin’s tubercle morphological variation among the Lodhas, one of the Particularly Vulnerable Group (s) (PVTGs) from Midnapore, West Bengal India quantifying parent-offspring transmission (father-daughter, mother-son) through pedigree analysis and helix measurements, to resolve familial aggregation and forensic applicability.
Materials and Methods
Pedigrees from 208 father-daughter and 208 father-son/mother-son pairs were obtained from the Lodhas, one of the Particularly Vulnerable Tribal Group(s) of Midnapore, Eastern India. A total of 416 biologically verified parent–offspring pairs were included, comprising 208 daughters and 208 sons. For each offspring, both paternal and maternal phenotypes were recorded, thereby allowing evaluation of father–daughter, mother–daughter, father–son, and mother–son transmission pathways Prior to then work it was explained to the participants for informed consent. Participants were recruited through household-based field surveys using structured pedigree interviews. Only biologically confirmed parent–offspring pairs (as reported by community records and verified through cross-household validation) were included. Adopted individuals and incomplete pedigrees were excluded. DT presence was assessed morphologically by visual inspection and calliper measurement of posterior helix prominence (>2mm projection). Binary coding: Yes (DT present), No (absent). Chi-square tests of independence were performed with α = 0.05 (df = 1). Odds ratios (OR) and 95% confidence intervals (CI) were calculated using the Wald method. Relative Risk (RR) was additionally computed for comparative interpretation of transmission strength.
Results and Discussion
Examinations on Table 1 and 2 revealed among daughters (n=208), Darwin’s tubercle was present in 79 (38.0%) and absent in 129 (62.0%). In the father–daughter pairing, 58 of 79 DT-positive daughters (73.4%) had DT-positive fathers, whereas only 21 of 129 DT-negative daughters (16.3%) had DT-positive fathers.
Analysis demonstrated significant association (Chi2 =35.12, df=1, p<0.001) and an odds ratio of approximately 14.2, indicating daughters with a DT-positive father were over fifteen times more likely to express the trait than those with a DT-negative father. In the mother–daughter pairing, 52 of 89 daughters with DT-positive mothers (58.4%) were DT-positive, compared with 37 of 119 daughters (22.7%) whose mothers were DT-negative, indicated significant association (Chi2 =28.45, df=1, p<0.001) and an odds ratio around 4.7. These findings show strong but asymmetrical parental effects, with paternal transmission to daughters appearing more pronounced than maternal transmission.
Among sons (n=208), 104 (50.0%) were DT-positive and 104 (50.0%) DT-negative (Table 3).
For the father–son relationship, 91 of 101 sons with DT-positive fathers (90.1%) expressed DT, compared with only 13 of 107 sons (12.1%) of DT-negative fathers, demonstrated significant association (Chi2=86.49, df=1, p<0.001) and an odds ratio of about 65.8. In the mother–son pairing, 72 of 100 sons with DT-positive mothers (72.0%) were DT-positive versus 32 of 108 sons (29.6%) of DT-negative mothers (Chi²=42.35, df=1, p<0.001), odds ratio approximately 6.11. Overall, sons showed significantly (p<0.001) stronger aggregation with parental phenotype than daughters, and paternal influence on sons was found to be particularly striking. These robust chi-square values and large odds ratios across both tables clearly support strong familial clustering of the trait within this Lodha PVTG community.
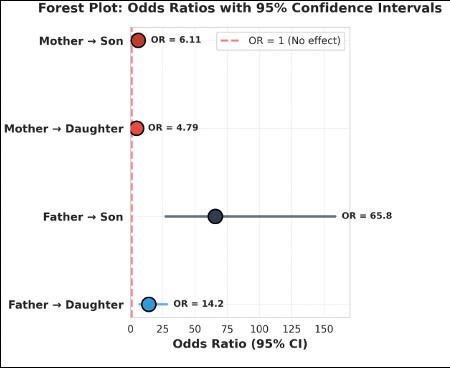
In addition to that computed forest plot (Figure 1) depicts odds ratios (ORs) with corresponding 95% confidence intervals (CIs) for parent–offspring transmission of the trait. All four transmission pathways demonstrated odds ratios substantially greater than unity, and in each case the 95% confidence intervals did not intersect the null value (OR = 1), indicating significant associations (p < 0.001). Maternal transmission showed moderate but significant effects, with sons of mothers with DT exhibiting higher odds of trait expression (OR = 6.11, 95% CI > 1) compared to daughters (OR = 4.79, 95% CI > 1) (Figure 2). Paternal transmission was considerably stronger, particularly in sons (OR = 65.8, 95% CI > 1), followed by daughters (OR = 14.2, 95% CI > 1). The confidence interval for the father–son pathway indicated relatively wide, and confirming a statistically robust and directionally consistent effect. Overall, the strength of association followed a clear gradient: Father → Son > Father → Daughter > Mother → Son > Mother → Daughter, demonstrating sex-specific differences in transmission intensity.
The significant (p <0.001) odds ratios observed across all parental pathways confirm the presence of strong familial aggregation for the trait, with a pronounced sex bias. From a forensic standpoint, the persistence of ORs significantly greater than 1—coupled with confidence intervals excluding the null—supports the reliability of the trait as a hereditary marker in kinship assessment. The exceptionally high odds associated with father–son transmission provide compelling evidence of sex-influenced inheritance, a pattern that enhances discriminatory power in pedigree reconstruction and disputed paternity scenarios.
The wider confidence interval associated with the father–son pathway, also indicated the magnitude of the effect and the clear separation from the null value strengthen inferential confidence. In forensic practice, traits demonstrating both high odds ratios and consistent directional inheritance are particularly valuable when used in conjunction with other morphological or genetic indicators.
Maternal transmission, although statistically significant, showed comparatively weaker effects, suggesting differential penetrance or expression modulated by parental sex and offspring sex. This hierarchical transmission pattern aligns with polygenic or epigenetically mediated models emphasizing the importance of probabilistic interpretation rather than deterministic assignment in forensic contexts. The findings on Darwin’s tubercle underscore its status as a morphologically minor yet biologically informative trait shaped by the interaction of evolutionary history, embryological development and genetic complexity.
Consistent with Darwin’s original interpretation, DT exemplifies the persistence of vestigial traits through shared developmental pathways rather than through direct adaptive significance (Darwin, 1871; Gould, 1980). Its variable expression across populations further reflects the role of developmental plasticity in shaping human morphological diversity.
The embryological timing of auricular development provides a plausible explanation for the observed variability and asymmetry of DT. The formation of the helix during weeks 5–8 of gestation involves finely coordinated growth of auricular hillocks, rendering this structure particularly susceptible to subtle developmental fluctuations (Sadler, 2019; Moore et al., 2020). Such perturbations may account for bilateral asymmetry and incomplete concordance among genetically identical individuals, as reported in twin studies (Porter, 2000).
Genetic evidence increasingly supports a polygenic threshold model for DT expression rather than a simple Mendelian mode of inheritance. High frequencies of DT among offspring of unaffected parents and approximately 20% discordance among monozygotic twins indicate substantial non-genetic modulation (McDonald, 2011). Moreover, observed parent–offspring concordance patterns—such as stronger father–daughter helix similarity and mother–son helix–tragus correspondence—suggest subtle sex-specific influences operating within a broader polygenic background (Al-Dulaimi et al., 2025). These observations align with genome-wide association studies demonstrating that craniofacial and auricular traits are influenced by multiple loci with small additive effects, further shaped by developmental and environmental factors (Liu et al., 2012; Claes et al., 2014).
Within an evolutionary–developmental framework, DT illustrates how vestigial structures are retained through conserved embryological pathways, supporting the concept of “descent with modification” central to evo-devo theory (Hall, 2003). Rather than being evolutionarily inconsequential, such traits provide insight into the developmental constraints and historical contingencies that shape human morphology.
From an applied perspective, the forensic and anthropological relevance of DT lies in its utility as a non-invasive morphological marker. Studies in forensic science have demonstrated that external ear features, when assessed systematically, contribute meaningfully to individual identification and ancestry estimation (Iannarelli, 1989; Meijerman et al., 2007).
In anthropologically diverse regions such as Eastern and North-Eastern India, DT can aid in reconstructing inheritance patterns and enhancing population differentiation when integrated with other somatotypic and craniofacial traits (Bury et al., 2016; McDonald, 2011). Thus, Darwin’s tubercle remains a valuable feature for understanding human variation at the intersection of evolution, development, and applied anthropology.
Conclusion
The results demonstrate strong familial aggregation of Darwin’s tubercle with pronounced sex bias. These findings are consistent with autosomal inheritance exhibiting sex-specific penetrance and developmental sensitivity. In addition to its population biological relevance, the observed aggregation patterns suggest that Darwin’s tubercle may provide supplementary probabilistic information in applied anthropological contexts, including forensic familial assessment, when used cautiously and in combination with other indicators. In summary, the statistically significant associations and sex-biased aggregation patterns indicate strong familial clustering within this population. However, given its polygenic and developmentally modulated nature, Darwin’s tubercle should be regarded as an informative but non-exclusive morphological marker in anthropological and forensic contexts. Future studies incorporating molecular validation would further clarify its evidentiary.
Acknowledgements
Authors are grateful to the participants and acknowledge the financial support of University of Calcutta [BI 65, (8 & 9)] and from UGC NET fellowship to PD.
References
Al-Dulaimi, R. K., Singh, P., & Verma, S. (2025). Inheritance patterns of auricular traits with special reference to Darwin’s tubercle. International Journal of Morphology, 43(1). (Advance online publication).
Bury, J., Łoś, M., & Kaczmarek, M. (2016). Morphological variation of the human auricle: Population and developmental perspectives. Anthropologischer Anzeiger, 73(3), 225–240. https://doi.org/10.1127/anthranz/2016/0643
Claes, P., Liberton, D. K., Daniels, K., Rosana, K. M., Quillen, E. E., Pearson, L. N., et al. (2014). Modeling 3D facial shape from DNA. PLoS Genetics, 10(3), e1004224. https://doi.org/10.1371/journal.pgen.1004224
Darwin, C. (1871). The descent of man, and selection in relation to sex (Vol. 1). London, UK: John Murray.
Deopa, D., Thakkar, H. K., Prakash, C., Niranjan, R., & Barua, M. P. (2013). Anthropometric measurements of external ear of medical students in Uttarakhand region. Journal of the Anatomical Society of India, 62(1), 79–83.
Gould, S. J. (1980). The panda’s thumb: More reflections in natural history. New York, NY: W. W. Norton & Company.
Hall, B. K. (2003). Descent with modification: The unity underlying homology and homoplasy as seen through evolution and development. Biological Reviews, 78(3), 409–433. https://doi.org/10.1017/S1464793102006097
Hrdlička, A. (1933). The external ear: Its variations and anomalies. American Journal of Physical Anthropology, 17(2), 249–266.
Iannarelli, A. V. (1989). Ear identification. Fremont, CA: Paramount Publishing.
Liu, F., van der Lijn, F., Schurmann, C., Zhu, G., Chakravarty, M. M., Hysi, P. G., et al. (2012). A genome-wide association study identifies genetic loci influencing facial morphology in Europeans. PLoS Genetics, 8(9), e1002932. https://doi.org/10.1371/journal.pgen.1002932
McDonald, J. H. (2011). Myths of human genetics. Baltimore, MD: Johns Hopkins University Press.
Meijerman, L., van der Lugt, C., & Maat, G. J. R. (2007). Cross-sectional anthropometric study of the external ear. Journal of Forensic Sciences, 52(2), 286–293. https://doi.org/10.1111/j.1556-4029.2006.00376.x
Moore, K. L., Persaud, T. V. N., & Torchia, M. G. (2020). The developing human: Clinically oriented embryology (11th ed.). Philadelphia, PA: Elsevier.
Murgod, V., Angadi, P., & Hallikerimath, S. (2013). Anthropometric study of the external ear in a South Indian population. Journal of Forensic and Legal Medicine, 20(5), 379–383. https://doi.org/10.1016/j.jflm.2013.02.004
Porter, C. J. (2000). Darwin’s tubercle: A vestigial feature of the human ear. Journal of Anatomy, 197(2), 317–320. https://doi.org/10.1046/j.1469-7580.2000.19720317.x
Sadler, T. W. (2019). Langman’s medical embryology (14th ed.). Philadelphia, PA: Wolters Kluwer.
Singh, P., & Purkait, R. (2009). Observations on external ear morphology in an Indian population. Anthropology, 47(1–2), 1–12.
Wood-Jones, F. (1931). The principles of anatomy as seen in the hand. London, UK: Baillière, Tindall & ox.

Received: Decembe 23rd, 2025;
Accepted: March 16th, 2026 ;
Online first: March 20th, 2026;
Published: TBA
Copyright: © 2025 Das & Bandyopadhyay. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.